|
We see in the figure below, cations increase in size when going down a group and they decrease in size when going across a period. If S gains two electrons, the electron configuration is and when F gains one electron, the electron configuration is. Īnions are larger than their parent atoms because when one or more electrons are added, the repulsions between electrons push them further from the nucleus. If Al loses three electrons to become Al 3+ ion, the electron configuration is. The Na + ion configuration is 1s 22s 22p 6 or. The electron configuration for the sodium atom is 1s 22s 22p 63s 1. For example, Na loses one electron from the 3s orbital, the valence shell, to form Na +. The electrons in the same shell do not shield well, therefore, we see a decrease in atomic radius as we go across a period.Ī cation is smaller than its parent atom. Recall that as electrons are being added so are protons. In Period 2, lithium has a +3 nuclear charge and fluorine has a +9 nuclear charge. 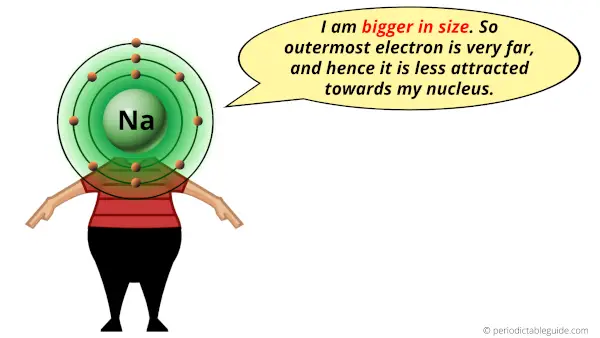
When going across a period, Z eff increases, therefore, the atomic radius decreases going across a period. For example, in Group 1A, Na has a valence shell of 3s while Rb has a valence shell of 5s. Going down a group on the periodic table successively larger valence shell oribtals are occupied by the electrons. We can see in the figure below that atomic radius decreases as we move across a period and increases as we move down a group on the periodic table. For the carbon-chlorine bond, the distance between the two nuclei is 99 pm + 77 pm = 176 pm. The atomic radius is measured as half the distance between the nuclei of two identical atoms that are bonded. However, we know we can not know the exact position of an electron or electrons in the outermost shell. Ideally, atomic radius would be measured from the nucleus of the atom to the electron(s) in the outermost shell. 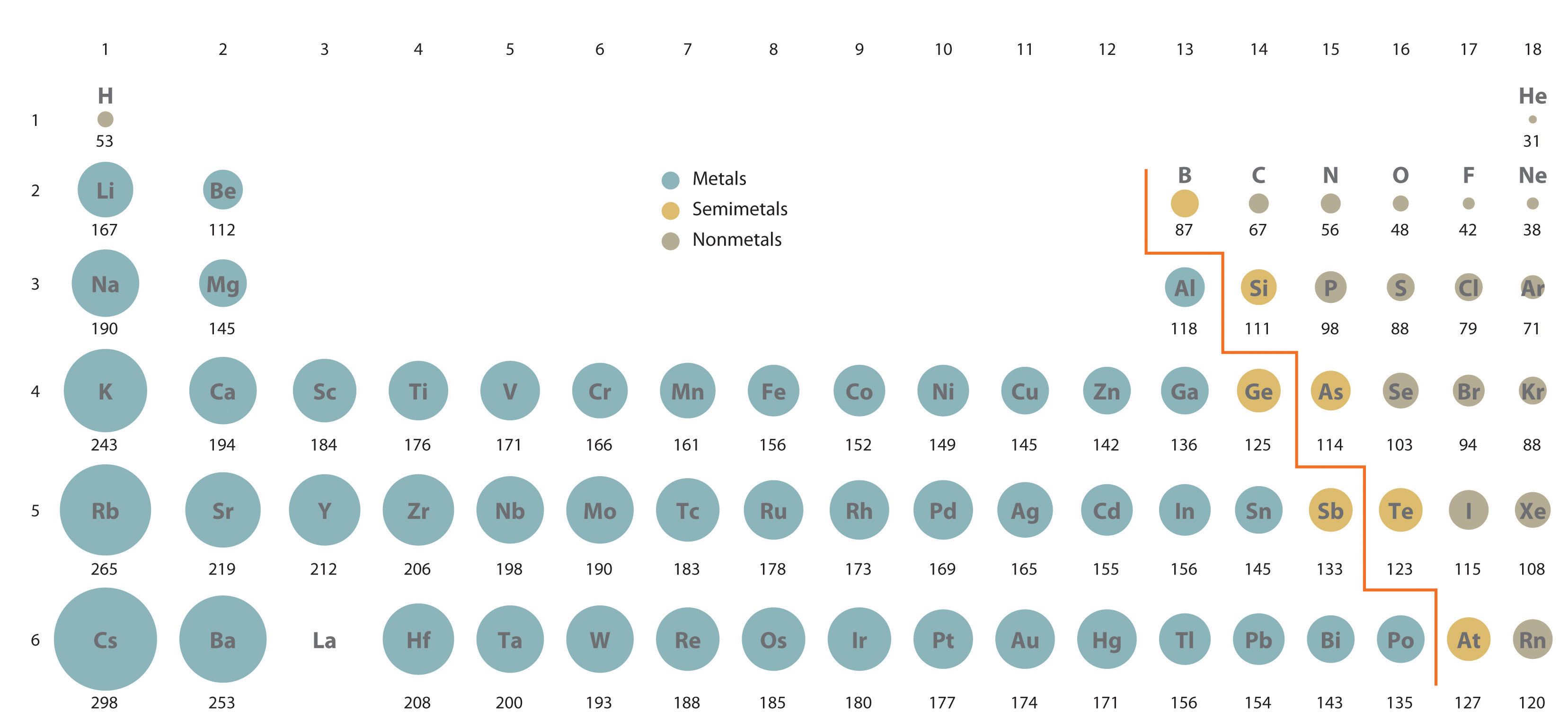 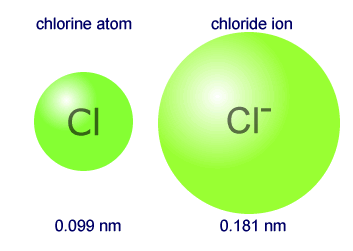
Here we discuss atomic and ionic radii and the periodic trends. We have already learned how to use the periodic table to write electron configurations and orbital box diagrams.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
